Blinded labeling is an essential element in all clinical trials. To remove the risk of bias, active pharmaceutical ingredient (API) drugs, delivery apparatus, and kits must be visually indistinguishable from placebo units — including their respective labels. The content and creation of these labels, though, is tightly regulated and benefits from the guidance of an experienced partner.
How Are GMP-Compliant Blinded Labels Created?
Sponsors must provide a blinded product for Phase 1B clinical trials and all subsequent trials. API and placebo labels’ content is determined by regulations in the nations hosting each trial site, but typically, each label shows: a batch number, a caution statement (indicating the unit is for clinical trial use only), a child warning (if the unit is for at-home use), storage conditions, and manner of administration. Some countries also require an expiration date or a retest date on the label, as well as emergency contact information.
Singota begins the process (Fig. 1) by determining the locations of a client’s trial sites. We commonly print in the U.S. and across the E.U., but we constantly add new locations (e.g., Australia). We can help clients by researching the blinded labeling regulations wherever their trial sites are located, though we also are experienced in working with larger clients’ regulatory departments, who do not require such assistance.
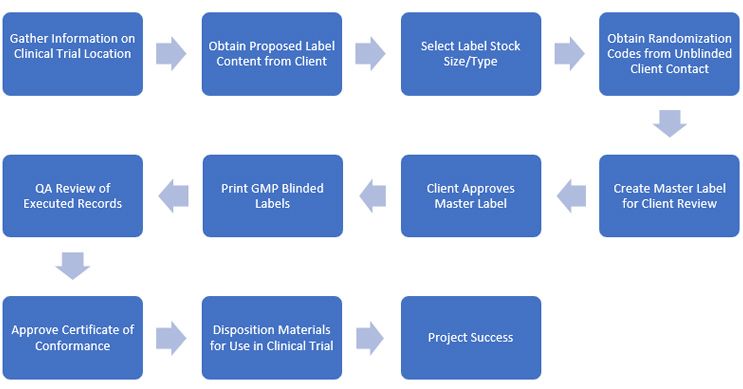
Fig. 1 — Singota’s blinded labeling flowchart
Next, we request the client identify their key players — who is blinded and who is unblinded — so no information that would unblind people is erroneously shared during the study. The client needs to identify at least two people (e.g., a main contact and a backup) who can receive our batch records, and with whom we can communicate freely (i.e., about which units are placebo and which are API).
Labeling clients also must identify who will be providing their blinded identifier numbers. There will exist two lists: unique numbers associated with the placebo and unique numbers associated with API units. In this case, “numbers” is a generic term; clients can choose any sequence of numbers/letters/symbols they like, and often secure a statistician to provide those identifiers. A contact on the client end also must confirm/certify that each number provided to the label maker is unique.
Many labeling clients do not realize going into a trial that the randomization list is not something a label and kitting provider can produce for them. Some companies have an in-house, unblinded statistician who can perform the task, while others outsource it to a third party. Once the randomization list is complete, the label maker can begin batch records, which allow for generation of the labels.
At Singota, each label number is entered manually and printed individually; a two-person team verifies the randomization code printed on each label. Ultimately, the same code number may be printed, for example, on five individual items within a dose kit, as well as a sixth label created for the kit itself. This is why verification redundancy is so important. When labels are applied to a product, each label will be applied by the performer and verified by a second person before being approved by a QA oversight team member.
Consider, too, that some countries require labels in two languages (e.g., Canada, Germany). In other cases, label stock is a factor. Leaflet stock (i.e., labels that unfold to show more information) may take longer to print. Certified temperature label stock (i.e., labels that will not lose adhesion stored at -80°C) or multicolor labels (e.g., a client wants to include a multicolor company logo) may also impact printing lead time.

In any scenario, Singota generates a master label (Fig. 2) so each client can approve its appearance for use on their product. Some clients also request photos of the assembled kits or have their own batch release paperwork. Once we have completed label production, the client has reviewed the records, and we have addressed any batch record changes, we are ready to move forward with dispositioning the batch. We fill out our certificate of compliance, the client approves it, and the kits can be dispositioned and shipped.
Streamlining Blinded Labeling
The earlier an organization engages a blinded labeling partner, the better. Earlier engagement allows the label maker more time to work out details and review documentation, including batch records. That said, Singota has the capability to serve “last-minute” projects, as well.
We combine deep experience in GMP-compliant blinded labeling with supply chain ability, so we can ship completed kits directly from our building. We also offer materials storage and, if necessary, relabeling. While Singota also offers manufacturing/aseptic filling, many of our labeling clients come to us as a third party to provide this specific service.
Moreover, we strive to help clients navigate the blinded labeling process as smoothly as possible, which can include sending the client randomization list examples, as well as examples of the label size and the information it must contain. Fundamentally, the promise we make is SISPQ — safety, identity, strength, purity, and quality — at all times. To learn more, visit us at Singota.com and follow us on LinkedIn.
About The Author

Beth Baxter is an Associate Quality Assurance Manager at Singota Solutions. She has been with the Company over eight years, during which she has served in scientist roles in the QC-Development laboratory and as a Senior Quality Specialist in the Quality Assurance department. Beth obtained her MS in analytical chemistry from Indiana University and is a member of the American Society for Quality.
About Singota Solutions
Singota Solutions is a contract development and manufacturing organization (CDMO) focused on helping clients in the pharmaceutical, animal health, and biotechnology industries move their products through the drug development pipeline faster by being agile, accountable, and transparent.